What Are the Different Ways to Represent Compounds Check All That Apply
three.3: Representing Compounds- Chemic Formulas and Molecular Models
- Page ID
- 37953
Chemistry is the experimental and theoretical study of materials on their properties at both the macroscopic and microscopic levels. Understanding the human relationship between backdrop and structures/bonding is also a hot pursuit. Chemistry is traditionally divided into organic and inorganic chemistry. The former is the written report of compounds containing at least one carbon-hydrogen bonds. By default, the chemical study of all other substances is called inorganic chemistry, a less well defined bailiwick.
However, the purlieus betwixt organic and inorganic compounds is not e'er well defined. For instance, oxalic acid, H2C2O4, is a chemical compound formed in plants, and it is generally considered an organic acrid, but it does non contain any C-H bond. Inorganic chemistry is besides closely related to other disciplines such every bit materials sciences, concrete chemistry, thermodynamics, globe sciences, mineralogy, crystallography, spectroscopy etc.
A chemical formula is a format used to limited the structure of atoms. The formula tells which elements and how many of each element are nowadays in a compound. Formulas are written using the elemental symbol of each cantlet and a subscript to denote the number of elements. This note tin be accredited to Swedish pharmacist Jons Jakob Berzeliu. The about common elements present in organic compounds are carbon, hydrogen, oxygen, and nitrogen. With carbon and hydrogen nowadays, other elements, such equally phosphorous, sulfur, silicon, and the halogens, may exist in organic compounds. Compounds that practice not pertain to this dominion are chosen inorganic compounds.
Molecular Geometry and Structural Formula
Agreement how atoms in a molecules are arranged and how they are bonded together is very important in giving the molecule its identity. Isomers are compounds in which 2 molecules tin can accept the same number of atoms, and thus the same molecular formula, but can have completely unlike physical and chemical properties because of differences in structural formula.
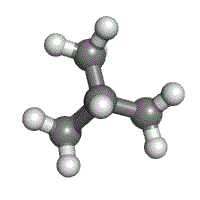

Thou ethylpropane and butane take the aforementioned molecular formula of C4Hx, simply are structurally different (methylpropane on the left, butane on the correct).
Polymers
A polymer is formed when pocket-sized molecules of identical structure, monomers, combine into a large cluster. The monomers are joined together past covalent bonds. When monomers repeat and bind, they form a polymer. While they can exist comprised of natural or constructed molecules, polymers often include plastics and rubber. When a molecule has more than 1 of these polymers, square parenthesis are used to show that all the elements within the polymer are multiplied by the subscript exterior of the parenthesis. The subscript (shown every bit n in the example below) denotes the number of monomers present in the macromolecule (or polymer).
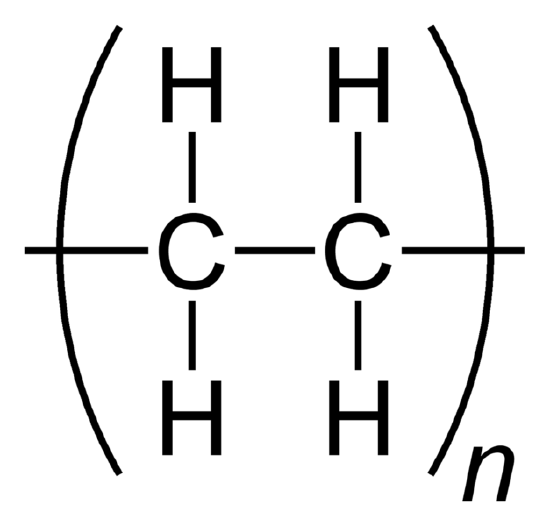
Ethylene becomes the polymer polyethylene.
Molecular Formula
The molecular formula is based on the bodily makeup of the compound. Although the molecular formula tin can sometimes be the same as the empirical formula, molecular compounds tend to be more helpful. Yet, they do not depict how the atoms are put together. Molecular compounds are too misleading when dealing with isomers, which take the aforementioned number and types of atoms (see to a higher place in molecular geometry and structural formula).
Ex. Molecular Formula for Ethanol: CtwoH6O.
Empirical Formula
An empirical formula shows the almost basic form of a compound. Empirical formulas prove the number of atoms of each element in a compound in the nearly simplified state using whole numbers. Empirical formulas tend to tell united states very lilliputian well-nigh a compound because i cannot determine the construction, shape, or properties of the compound without knowing the molecular formula. Usefulness of the empirical formula is decreased because many chemical compounds can have the same empirical formula.
Ex. Discover the empirical formula for C viii H 16 O 2 .
Answer: C4H 8 O (dissever all subscripts by 2 to get the smallest, whole number ratio).
Structural Formula
A structural formula displays the atoms of the molecule in the club they are bonded. It also depicts how the atoms are bonded to ane another, for case single, double, and triple covalent bail. Covalent bonds are shown using lines. The number of dashes indicate whether the bond is a single, double, or triple covalent bail. Structural formulas are helpful because they explain the properties and structure of the compound which empirical and molecular formulas cannot always correspond.
Ex. Structural Formula for Ethanol: 
Condensed Structural Formula
Condensed structural formulas show the order of atoms similar a structural formula simply are written in a single line to relieve space and get in more convenient and faster to write out. Condensed structural formulas are also helpful when showing that a group of atoms is continued to a single atom in a compound. When this happens, parenthesis are used around the group of atoms to evidence they are together.
Ex. Condensed Structural Formula for Ethanol: CHthreeCH2OH (Molecular Formula for Ethanol CtwoH6O).
Line-Angle Formula
Because organic compounds tin be complex at times, line-angle formulas are used to write carbon and hydrogen atoms more efficiently by replacing the letters with lines. A carbon atom is present wherever a line intersects some other line. Hydrogen atoms are then assumed to complete each of carbon's four bonds. All other atoms that are connected to carbon atoms are written out. Line angle formulas help show structure and order of the atoms in a compound making the advantages and disadvantages similar to structural formulas.
Ex. Line-Angle Formula for Ethanol: 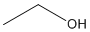
Formulas of Inorganic Compounds
Inorganic compounds are typically not of biological origin. Inorganic compounds are made up of atoms connected using ionic bonds. These inorganic compounds can exist binary compounds, binary acids, or polyatomic ions.
Binary compounds
Binary compounds are formed between two elements, either a metallic paired with a nonmetal or ii nonmetals paired together. When a metal is paired with a nonmetal, they class ionic compounds in which one is a negatively charged ion and the other is positvely charged. The net accuse of the compound must then become neutral. Transition metals have different charges; therefore, it is important to specify what type of ion it is during the naming of the compound. When two nonmetals are paired together, the compound is a molecular compound. When writing out the formula, the element with a positive oxidation land is placed first.
Ex. Ionic Compound: BaBr2(Barium Bromide)
Ex. Molecular Compound: NorthiiO4 (Dinitrogen Tetroxide)
Binary acids
Binary acids are binary compounds in which hydrogen bonds with a nonmetal forming an acid. However, there are exceptions such as NHiii, which is a base. This is because it shows no tendency to produce a H+. Because hydrogen is positively charged, information technology is placed kickoff when writing out these binary acids.
Ex. HBr (Hydrobromic Acrid)
Polyatomic ions
Polyatomic ions is formed when two or more atoms are continued with covalent bonds. Cations are ions that have are postively charged, while anions are negatively charged ions. The almost common polyatomic ions that exists are those of anions. The two main polyatomic cations are Ammonium and Mercury (I). Many polyatomic ions are typically paired with metals using ionic bonds to form chemical compounds.
Ex. MnO4 - (Polyatomic ion); NaMnOfour (Chemical Compound)
Oxoacids
Many acids have three dissimilar elements to form ternary compounds. When ane of those three elements is oxygen, the acid is known as a oxoacid. In other words, oxacids are compounds that contain hydrogen, oxgygen, and i other element.
Ex. HNO3 (Nitric Acid)
Complex Compounds
Certain compounds can announced in multiple forms yet hateful the same thing. A common example is hydrates: h2o molecules bond to another compound or element. When this happens, a dot is shown between H2O and the other function of the chemical compound. Because the H2O molecules are embedded within the compound, the compound is not necessarily "wet". When hydrates are heated, the water in the compound evaporates and the chemical compound becomes anhydrous. These compounds tin be used to attract h2o such as CoCl2. When CoCl2 is dry, CoClii is a blueish color wherease the hexahydrate (written beneath) is pinkish in colour.
Ex. CoClii · vi H2O
Formulas of Organic Compounds
Organic compounds contain a combination carbon and hydrogen or carbon and hydrogen with nitrogen and a few other elements, such equally phosphorous, sulfur, silicon, and the halogens. Nearly organic compounds are seen in biological origin, as they are found in nature.
Hydrocarbons
Hydrocarbons are compounds that consist of only carbon and hydrogen atoms. Hydrocarbons that are bonded together with merely single bonds are alkanes. The simplest example is methane (shown below). When hydrocarbons have ane or more double bonds, they are called alkenes. The simplest alkene is Ethene (C2H4) which contains a double bond between the ii carbon atoms.
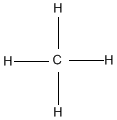
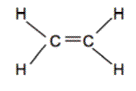
Ex. Methane on left, Ethene on right
Functional Groups
Functional groups are atoms continued to carbon chains or rings of organic molecules. Compounds that are within a functional grouping tend to have similar properties and characteristics. 2 common functional groups are hydroxyl groups and carboxyl groups. Hydroxyl groups terminate in -OH and are alcohols. Carboxyl groups end in -COOH, making compounds containing -COOH carboxylic acids. Functional groups also help with nomenclature past using prefixes to help proper noun the compounds that take like chemical properties.
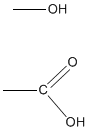
Ex. Hydroxyl Group on top; Carboxyl Group on bottom
References
- Miessler, Gary L. Inorganic Chemistry. 2nd. Upper Saddle River: Prentince Hall, 1999.
- Munowitz, Michael. Principles of Chemistry. Norton & Company: New York, 2000.
- Pettrucci, Ralph H. General Chemistry: Principles and Modern Applications. 9th. Upper Saddle River: Pearson Prentice Hall, 2007.
Issues
- Which of the post-obit formulas are organic?
- HClO
- C5H10
- CO2
- What is the name of the following formula?
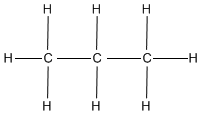
- Classify the following formulas into their appropriate functional group
- Acetic acid
- Butanol
- Oxalic acrid
- What are the empirical formulas for the following compounds?
- C12H10O6
- CHiiiCH2CH2CHtwoCHtwoCH2CHthree
- HthreeO
- What is the proper noun of the following figure and what is the molecular formula of the post-obit figure?
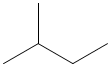
Answer Key:
1. b and c. 2. Propane. 3. a. carboxyl group, b. hydroxyl grouping, c. carboxyl grouping. 4. a. Chalf-dozenH5Othree, b. CviiH16, c. H3O. five. Methylbutane, C5H12
Contributors and Attributions
- Jean Kim (UCD), Kristina Bonnett (UCD)
Source: https://chem.libretexts.org/Bookshelves/General_Chemistry/Map:_A_Molecular_Approach_%28Tro%29/03:_Molecules_Compounds_and_Chemical_Equations/3.03:_Representing_Compounds-_Chemical_Formulas_and_Molecular_Models
0 Response to "What Are the Different Ways to Represent Compounds Check All That Apply"
Post a Comment